What is Pyrite?

Pyrite is a beautiful mineral that naturally forms into cubic crystal structures. As seen in the photo above, pyrite can become amazingly cuboidal, nearly perfect. These natural formations look artificial, almost alien in their perfection, yet they are simply the product of explainable geological events which occur all over the world.
Pyrite has the chemical formula FeS2, meaning a single iron atom is bonded to two sulfur atoms. This gives pyrite the capability to form simple cuboidal crystals, under the right environmental conditions. Further, it makes pyrite and important commercial source for both iron and sulfur. Many industries use pyrite to extract the iron, or they can use pyrite to produce sulfuric acid and other sulfur-based chemicals used for making paper and other industrial applications.
History of Pyrite
Historically, pyrite has been a mineral of human interest since the dawn of time. As long ago as 300 BC, pyrite was used in religious ceremonies for its perceived magical properties. The Romans used pyrites in a variety of artistic ways, such as using pyrite cubes and slivers as tiles in complex mosaics, such as the one seen below. In Medieval times, pyrite cubes were even used as medicine and patients were encouraged to swallow them whole. However, this is obviously no longer recommended because of the obvious damage it could do to your digestive tract.

More recently, pyrite was an important component in the production of the first firearms. These firearms were operated with pyrite used to create a spark. Commonly called “flintlock” rifles, these weapons drastically changed warfare and society in general. But pyrite did not stop there. During the gold rush of the 1800s, pyrite earned the moniker “fools gold” because its superficial resemblance to gold ended up confusing many novice gold miners.
Today, pyrite is still used in a number of commercial applications. Some battery companies use pyrite as a cathode within their batteries. Pyrite is a powerful electrical conductor and can pull electrons from the surrounding solution and direct them into a circuit. This property of pyrite has also been found to be useful within solar cells. Here, pyrite is formed into thin sheets which help collect the energy of the sun and convert it into usable electricity.
How is Pyrite Formed?
Pyrite is the end product of millions and billions of years of metamorphism and subsequent interactions of the rocks with dissolved substances within subsurface water aquifers. The formation of pyrite starts with chlorite iron deposits within the ground. These compounds were formed after igneous rocks are pressurized, heated, and subsequently changed over millions of years.
These chlorite compounds contain the iron found in pyrite, but they also contain silicone and oxygen, which must be removed and replaced with sulfur. For this to happen, conditions must be just right. Typically, these compounds are found under sandstone deposits, through which water aquifers run. This water carries dissolved sulfuric acid, which reaches down into the chlorite layers.
The reactions that ensue slowly replace the silicone and oxygen whithin these compounds with hydrogen. A byproduct of the reaction is the formation of quartz, which is a combination of silicone and oxygen. For this reason, thin layers of quartz are often found on the surface of pyrite specimens. Although it is only a few micrometers thick, it gives pyrite the fantastic luster and color that give it that fool’s gold look.
However, pyrite does not always form large, perfect cubes. In fact, in the majority of places pyrite is found it is in the form of small cubes, flakes, or even a shiny gold surface on certain fossils. This can be seen within the chambers of the fossilized shell below:

Why Does Pyrite form Cubes?
While bits of pyrite can form wherever an aquifer interacts with the metamorphic stones below it, there is a chance of pyrite forming. However, under the perfect conditions that persist for a long time, huge chunks of pyrite can be formed. These cubes can be up to 15 centimeters or more along one side. Pyrite can form these large, perfect cubes because it exhibits a cubic crystal system.
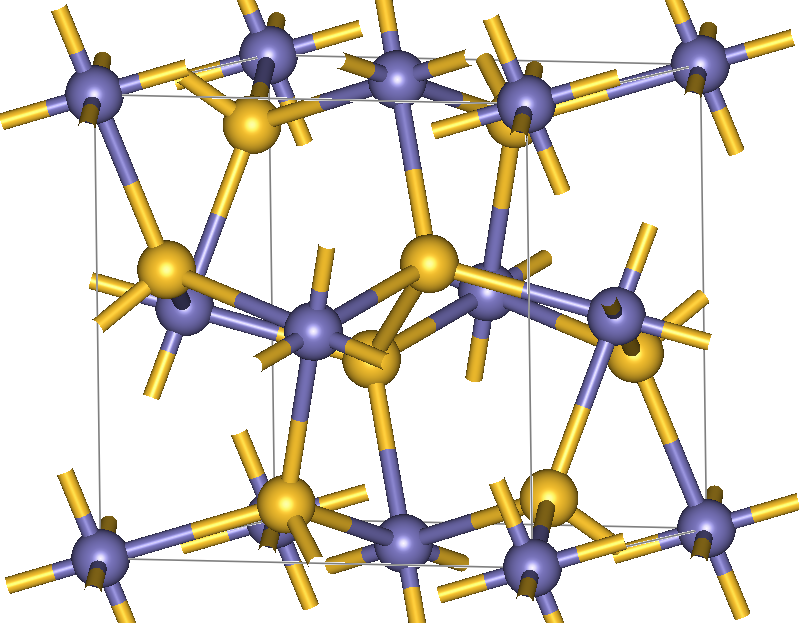
This perfect cubic crystal system can be seen in the image above. The yellow nodes represent the sulfur, while the iron atoms are purple. Together, these form a latticework with bonding points only available at the corners of the structure. Thus, as more chlorite containing rocks are converted to pyrite, the cubes grow in a structured and predictable pattern.
Under other conditions, pyrite can form other shapes such as disks, flat sheets, and less impressive crystal structures. For instance, below is a pyrite formation from Nevada. This formation, created by very different conditions than those that create cubes, forms thin strands of pyrite that overlap like quills on a porcupine.

What is so special about Pyrite from Navajun, Spain?
Navajun, Spain is an area where the conditions for forming pyrite have been perfect for millions of years. Navajun is located within the Cameros Basin, on the eastern side. In this basin, a perfect mixture of aquifers settles above rocks which are subject to low-grade hydrothermal metamorphism. In simpler terms, there is just the right amount of pressure, minerals, and water to produce massive pyrite crystals, such as the one seen below.

Though pyrite can be found all over the world, the mines in Navajun produce some of the biggest and most intricate pyrite clusters available. Sometimes, the natural clusters are so big and interconnected that it seems only an abstract artist could have imagined them. They are highly aesthetic, sharp, lustrous, and nearly perfect.
More than this, the pyrite deposits within the eastern Cameros Basin are thought to be as dense as 100 kg per cubed meter. This makes Navajun, Spain one of the most important and prolific pyrite mines in the world. Further, unlike in some parts of the world, the pyrite here is nearly all cuboidal and has already been exposed through the efforts of previous miners.
Interested in Mining Pyrite?
If you don’t believe that these amazing structures actually come out of the ground, you can go see it for yourself. Piritas de Navajun is a pyrite mine in Navajun which is open to the public. After scheduling a mining expedition on their website, customers are allowed to explore the mine, dig for specimens, and take what they find. You might be lucky enough to find massive, perfectly cubed specimens grouped in aesthetically pleasing clusters. But be sure to bring an extra suitcase. Pyrite is heavy!

Don’t Want to Get Dirty?
That’s fine too! Pyrite clusters are widely available through collectors and mineral dealers. These clusters have also been cleaned, and any loose pyrite cubes glued securely in place. This preserves the natural beauty of the clusters in the original formation they were found in, but without the surrounding rock. These clusters can be magnificent, showing the random but interconnected nature of crystal formations.


